VGR-R01 injection, one of the proprietary core products of Shanghai Vitalgen BioPharma Co., Ltd. (hereinafter referred to as “Vitalgen”), was granted implied CTA approval by the Center for Drug Evaluation of the NMPA for the treatment of Bietti crystalline dystrophy (BCD) on November 1, 2022, which is the world's first therapeutic product approved for clinical trial. On September 21, 2023, the surgical administration of all subjects was successfully completed in the Phase I/II clinical trial of VGR-R01, marking another critical step toward the marketing of this world-first therapeutic product.
Under the leadership of the principal investigators Professor Wenbin Wei from Beijing Tongren Hospital, Capital Medical University, and Professor Xiaodong Sun from the Shanghai General Hospital, the safety and efficacy clinical study of VGR-R01 was officially initiated in March 2023; and the enrollment and administration of VGR-R01 to all subjects were completed in half a year. So far, the Phase I/II clinical trial of VGR-R01 has yielded encouraging data: all subjects showed good safety and tolerability, and no adverse events (AE) possibly related to VGR-R01 was reported. The preliminary efficacy data showed that VGR-R01 could rapidly reverse or control disease progression in all dose groups in the dose escalation phase, enabling BCD patients with an average course of more than 10 years to improve the best corrected visual acuity (BCVA) or functional vision in dim conditions (Modified Rivermead Mobility Index, MRMI), thereby improving the quality of life (VFQ-25 score). The Vitalgen project team plans to discuss the further development and approval plan of the product with regulatory authority in the near future, and strive to promote the faster approval of the product to help BCD patients out of the dilemma of no cure for medical.

Figure 1 Investigators from Beijing Tongren Hospital, Capital Medical University
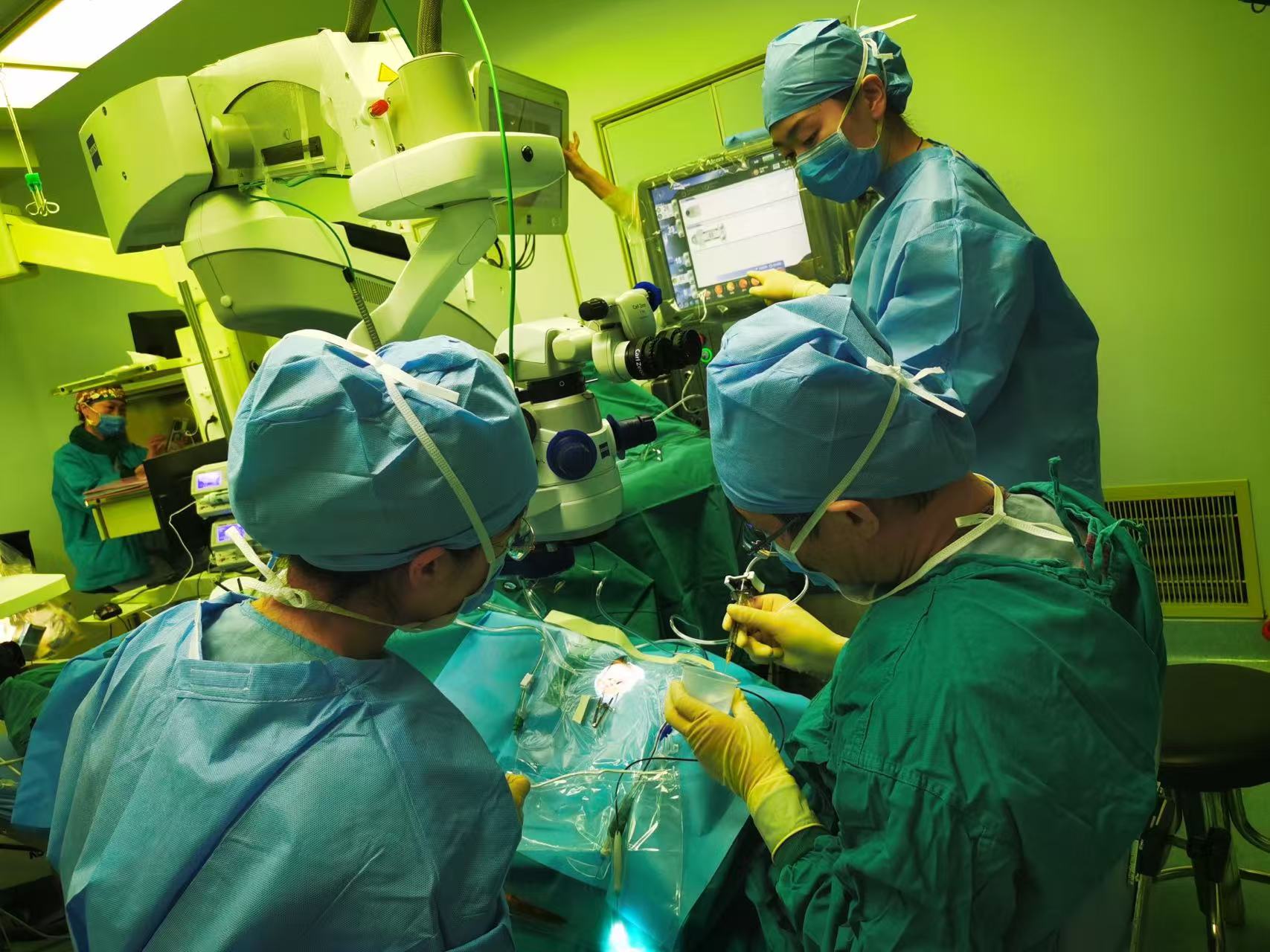
Figure 2 A BCD patient is receiving a surgery in Beijing Tongren Hospital, Capital Medical University
Dr. Xiaoping Zhao, CEO of Vitalgen, said: “I would like to thank all the BCD patients who participated in this study, the Suzhou Aitong Public Service Center (an organization of patients with retinal pigment degeneration), and all the investigators from Beijing and Shanghai study sites for your cooperation with Vitalgen to achieve another major milestone in BCD treatment.
So long as all God's children be
Relieved from hunger, free from cold
It cares not if, from wooded mountains
It comes to vast sufferings untold.”
As a rare disease with a high incidence in East Asian populations, BCD is not concerned by European and American pharmaceutical companies, and our local enterprises need to shoulder the heavy responsibility. The original intention of the establishment of Vitalgen is to commit to making all diseases have the chance to cure, and develop a true global FIC gene therapy product. VGR-R01 best explains Vitalgen’s positioning: starting from the clinical needs of Chinese patients, design the gene therapy product, establish the disease animal model, and finally return to the clinical treatment, reflecting the original intention of the Vitalgen team.
Vitalgen will continue to efficiently promote the follow-up clinical research of VGR-R01, looking forward to contributing important breakthroughs in China's independent innovation in the biotechnology industrial revolution, and bringing light to many BCD patients. “
About BCD
Bietti crystalline dystrophy (BCD), also known as Bietti crystalline retinopathy, is an autosomal recessive, progressive retinal degenerative disease. The pathogenic gene for BCD is CYP4V2 located at 4q35, which encodes a protease involved in lipid metabolism. BCD is found worldwide, but more prevalent in Chinese, Japanese and Korean populations. Most patients with BCD develop symptoms such as night blindness and decreased visual acuity in their 20s and 40s, and develop legal blindness in their 50s and 60s. BCD can be diagnosed based on clinical characteristics and genetic testing to identify the biallelic pathogenic variation in CYP4V2, but there is still no effective clinical treatment option available.
About VGR-R01
VGR-R01 is a gene therapy product for patients with BCD caused by CYP4V2 gene mutation. CYP4V2 protein is a member of the P450 enzyme family. It is highly expressed in retinal pigment epithelium (RPE), with fatty acid hydroxylase activity, and is related to lipid metabolism. Mechanism of action of VGR-R01 is gene replacement.
After VGR-R01 is administered via subretinal injection, AAV capsid protein mediates transduction of RPE cells, delivering the VGR-R01 gene expression cassette to the nucleus. The VGR-R01 expression cassette exists as free DNA and expresses CYP4V2 protein in RPE cells to reconstitute the fatty acid hydroxylase activity of the cells. VGR-R01 aims to prevent or improve the structural and/or functional damage of RPE cells, photoreceptor cells and choroid by correcting the fatty acid metabolism disorder in the retina of patients, so as to correct visual impairment, protect residual visual function, or delay vision deterioration.

Figure3 Mechanism of action of VGR-R01