Shanghai Vitalgen BioPharma Co., Ltd. (Vitalgen) has unveiled a groundbreaking treatment that promises to transform the lives of individuals suffering from Bietti Crystalline Dystrophy (BCD). Developed in-house, their revolutionary VGR-R01 injection has successfully completed Phase I/II clinical trials by September, 2023, marking a significant milestone as the world’s first IND-approved and clinically tested therapy for this intended indication. Leading the charge in this crucial research are Professor Wei Wenbin from Beijing Tongren Hospital, CMU, and Professor Sun Xiaodong from Shanghai General Hospital. Recently, the remarkable progress achieved by Vitalgen’s VGR-R01 injection, from animal models to intended indications and molecular breakthroughs, has garnered widespread attention. Its outstanding clinical efficacy has earned it a dedicated feature on Beijing TV’s renowned show, “Good Morning Beijing”.
One of the clinical trial participants expressed their heartfelt sentiment, stating, “After more than a decade since my BCD diagnosis, having a targeted gene therapy that aligns with our specific needs is nothing short of a miracle. Many of my fellow BCD sufferers, who were unable to participate in clinical trials, are anxiously awaiting this treatment. The relentless progression of this disease is irreversible, and even a common cold can dramatically impair our vision. The prolonged wait for a cure can shatter our hopes. That’s why BCD patients worldwide are eagerly anticipating the earlier release of Vitalgen’s VGR-R01 injection.”
Professor Li Wei, the visionary founder of Vitalgen, emphasized, “BCD, a hereditary retinal disease that has received limited attention from Western pharmaceutical giants, is a prevalent RP subtype among East Asian populations. In line with the guiding principle of ‘Solving China’s Problems with Chinese Solutions,’ our team at the Institute of Zoology, Chinese Academy of Sciences, leveraged cutting-edge gene modification tools with proprietary intellectual property rights to achieve groundbreaking advancements in BCD animal models and experiments. Through a highly efficient collaboration with Vitalgen and Professor Wei’s esteemed team, BCD gene therapy has rapidly transitioned into a game-changing ‘new productivity’, offering a glimmer of hope to previously untreatable BCD patients.”
Professor Wei Wenbin, renowned for his expertise at Beijing Tongren Hospital, CMU, shared his perspective, saying, “Throughout our extensive clinical experience, our team has witnessed the struggles of countless individuals afflicted with Bietti Crystalline Dystrophy. We have steadfastly followed the progress of theories and technologies related to this disease. Through our collaboration with Professor Li Wei and the Vitalgen team, this globally innovative BCD gene therapy has demonstrated promising initial efficacy in our trial participants. We are overjoyed to witness significant clinical improvements in the vision of most patients. For instance, one patient, who previously couldn’t discern any letters on an eye chart, now scores 40 letters and is capable of independently traveling to Beijing for follow-up appointments and even embarking on adventurous outings like climbing the Great Wall. This signifies a life-changing opportunity for middle-aged individuals to reenter the workforce and support their children through their own labor.”
Dr. Zhao Xiaoping, co-founder and chairman of Vitalgen, underlined, “Though BCD is categorized as a rare disease with relatively low prevalence, the overall number of affected individuals remains significant. For thousands of families, the prime working years of their loved ones have been overshadowed by legal blindness caused by BCD, imposing an enormous burden. The successful development of the VGR-R01 injection serves as a testament to the collaborative innovation ecosystem, where government, industry, academia, and research synergistically contribute. Thanks to the supportive gene therapy-related policies in China and our unwavering commitment to addressing the urgent clinical needs of Chinese patients, we have pioneered independent technological innovations and catalyzed the industrialization of domestic biopharmaceutical companies. Ultimately, this breakthrough will bring immense benefits to BCD patients across China.”
About BCD
Bietti crystalline dystrophy (BCD), also known as Bietti crystalline retinopathy, is an autosomal recessive, progressive retinal degenerative disease. The pathogenic gene for BCD is CYP4V2 located at 4q35, which encodes a protease involved in lipid metabolism. BCD is found worldwide, but more prevalent in Chinese, Japanese and Korean populations. Most patients with BCD develop symptoms such as night blindness and decreased visual acuity in their 20s and 40s, and develop legal blindness in their 50s and 60s. BCD can be diagnosed based on clinical characteristics and genetic testing to identify the biallelic pathogenic variation in CYP4V2, but there is still no effective clinical treatment option available.
About VGR-R01
VGR-R01 is a gene therapy product for patients with BCD caused by CYP4V2 gene mutation. CYP4V2 protein is a member of the P450 enzyme family. It is highly expressed in retinal pigment epithelium (RPE), with fatty acid hydroxylase activity, and is related to lipid metabolism. Mechanism of action of VGR-R01 is gene replacement.
After VGR-R01 is administered via subretinal injection, AAV capsid protein mediates transduction of RPE cells, delivering the VGR-R01 gene expression cassette to the nucleus. The VGR-R01 expression cassette exists as free DNA and expresses CYP4V2 protein in RPE cells to reconstitute the fatty acid hydroxylase activity of the cells. VGR-R01 aims to prevent or improve the structural and/or functional damage of RPE cells, photoreceptor cells and choroid by correcting the fatty acid metabolism disorder in the retina of patients, so as to correct visual impairment, protect residual visual function, or delay vision deterioration.
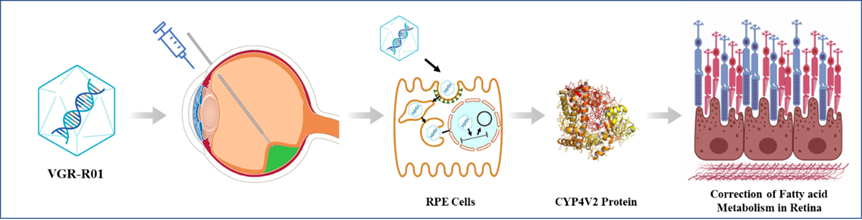
Figure Mechanism of action of VGR-R01