On May 9, 2024, Shanghai Vitalgen BioPharma Co., Ltd. (hereinafter referred to as “Vitalgen”) will globally publish the updated data on the phase I/II clinical trial of gene therapy product VGR-R01 in patients with Bietti crystalline dystrophy (BCD) in oral presentation form for the first time in the 27th Annual Meeting of American Society for Gene & Cell Therapy (ASGCT) to be held in Baltimore, fully demonstrating its excellent efficacy and safety advantages.
VGR-R01 is the world's first therapeutic product for BCD to receive clinical approve. Clinical data demonstrate that subretinal injection of VGR-R01 can lead to clinically significant improvement in visual acuity or enhancing functional vision in low-light environments, achieving the clinical effect of correcting visual impairment or protecting residual visual function, and is expected to render BCD no longer untreatable.

Detailed Data
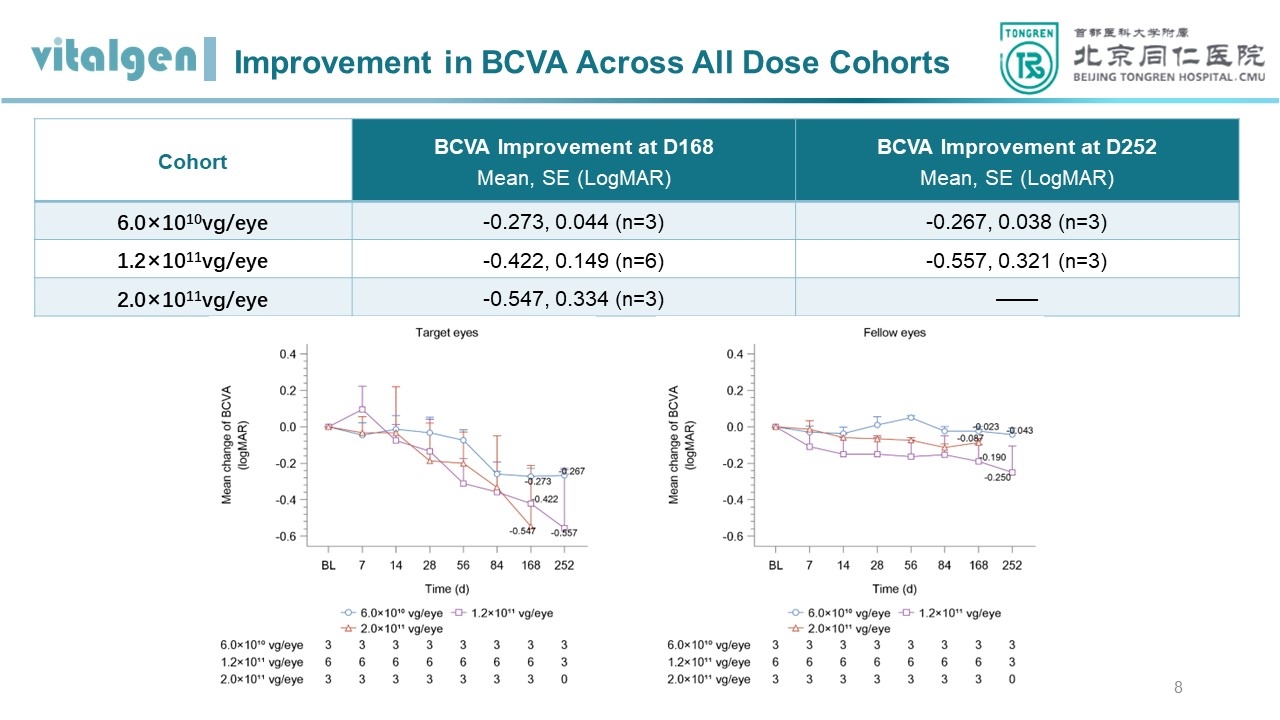
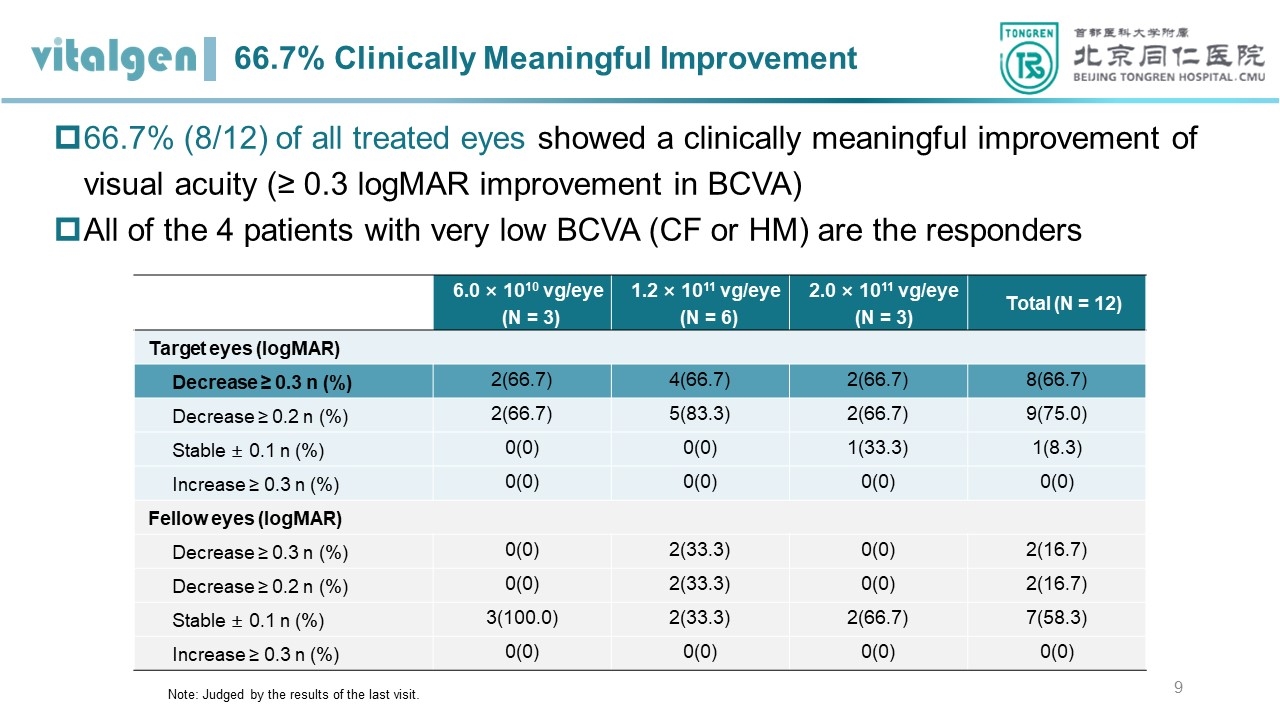
Improvement in visual acuity: As of March, 2024, in the low-, medium-, and high-dose groups of the study, the BCVA of the target eyes was improved by 0.267, 0.422, and 0.547 logMAR compared to baseline (equivalent to an increase of 13, 21, and 27 ETDRS letters) at the 9-month and 6-month follow-up, respectively. Moreover, 66.7% (8/12) of subjects showed a significant improvement in BCVA of the target eyes, i.e., a decrease of ≥ 0.3 log MAR (equivalent to an increase of 15 ETDRS letters). Among them, the baseline BCVA of 4 subjects (2 each in the medium- and high-dose groups) was either counting fingers (CF, 1.9 log MAR) or hand motion (HM, 2.3 log MAR) in the remaining, which showed significant improvements, i.e. equivalent to an increase of up to 40 letters, after VGR-R01 administration.
Improvement in functional visual acuity: The mean change in MLMT score of the target eyes increased by approximately 1~2 points in the low-, medium-, and high-dose groups. 41.7% (5/12) of subjects had a significant improvement, i.e., an increase of ≥ 2 points, suggesting the benefits of functional visual acuity in dim environments.
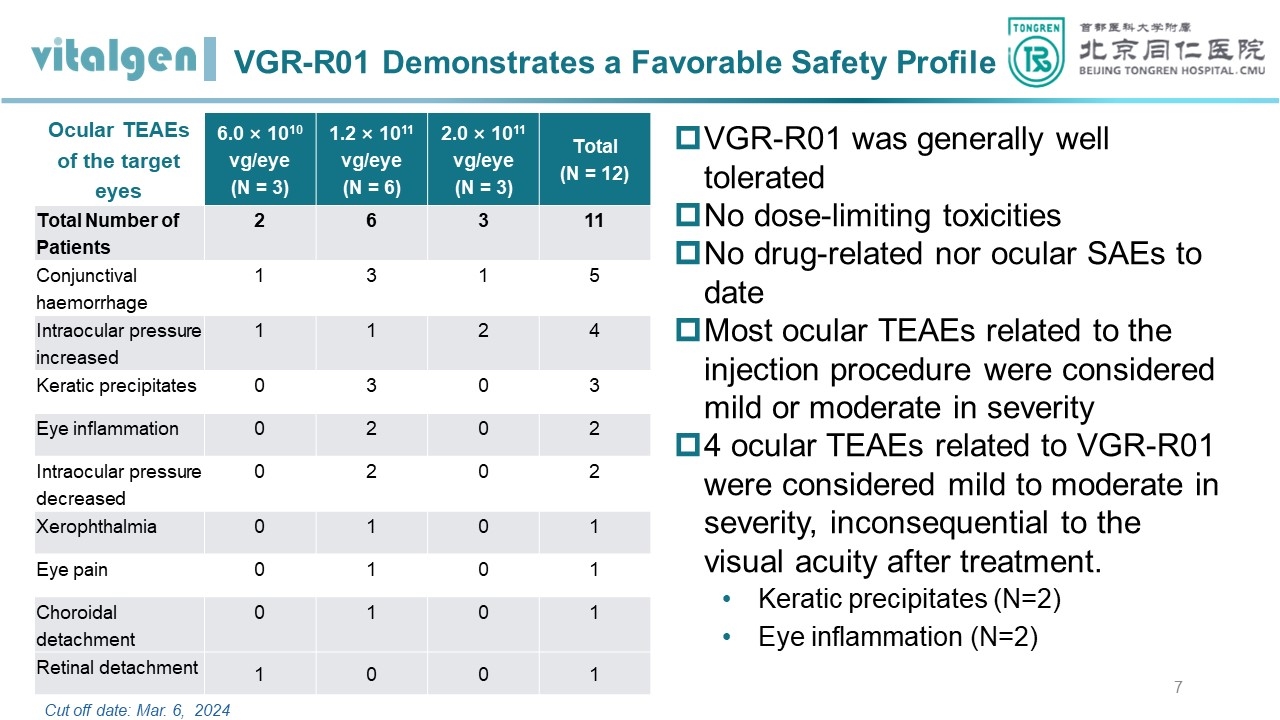
The safety data showed that VGR-R01 is generally well-tolerated and safe. There were no dose-limiting toxicities or drug-related SAEs; 4 subjects had eye inflammation that was assessed by the investigator to be possibly related to VGR-R01, with severity ranging from grade 1 to 2, which were all inconsequential to the visual acuity after treatment.


Excerpt of oral presentation
About BCD
Bietti crystalline dystrophy (BCD), also known as Bietti crystalline retinopathy, is an autosomal recessive, progressive retinal degenerative disease. The pathogenic gene for BCD is CYP4V2 located at 4q35, which encodes a protease involved in lipid metabolism. BCD is found worldwide, but more prevalent in Chinese, Japanese and Korean populations. Most patients with BCD develop symptoms such as night blindness and decreased visual acuity in their 20s and 40s, and develop legal blindness in their 50s and 60s. BCD can be diagnosed based on clinical characteristics and genetic testing to identify the biallelic pathogenic variation in CYP4V2, but there is still no effective clinical treatment option available.
About VGR-R01
VGR-R01 is a gene therapy product for patients with BCD caused by CYP4V2 gene mutation. CYP4V2 protein is a member of the P450 enzyme family. It is highly expressed in retinal pigment epithelium (RPE), with fatty acid hydroxylase activity, and is related to lipid metabolism. Mechanism of action of VGR-R01 is gene replacement.
After VGR-R01 is administered via subretinal injection, AAV capsid protein mediates transduction of RPE cells, delivering the VGR-R01 gene expression cassette to the nucleus. The VGR-R01 expression cassette exists as free DNA and expresses CYP4V2 protein in RPE cells to reconstitute the fatty acid hydroxylase activity of the cells. VGR-R01 aims to prevent or improve the structural and/or functional damage of RPE cells, photoreceptor cells and choroid by correcting the fatty acid metabolism disorder in the retina of patients, so as to correct visual impairment, protect residual visual function, or delay vision deterioration.
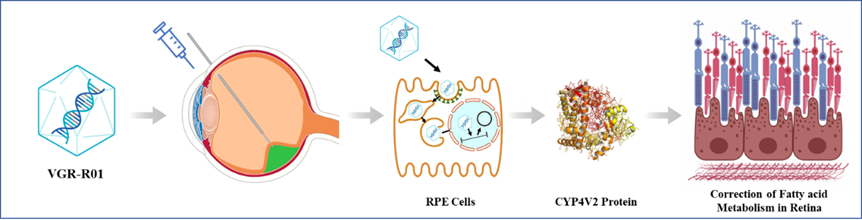
Figure Mechanism of action of VGR-R01